| World Journal of Nephrology and Urology, ISSN 1927-1239 print, 1927-1247 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Nephrol Urol and Elmer Press Inc |
| Journal website http://www.wjnu.org |
Original Article
Volume 9, Number 1, March 2020, pages 11-14
Are Inflammatory Markers Useful in Predicting Urinary Tract Infection After Transrectal Ultrasound-Guided Biopsy of the Prostate?
Nassib Abou Heidara, c, Muhammad Shahaitb, c, Aline Yaacoubiana, Rami Nasra, d
aDivision of Urology, Department of Surgery, American University of Beirut Medical Center, 11-0236/D50, Riad El-Solh 1107 2020, Beirut, Lebanon
bDepartment of Urology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA
cBoth authors contributed equally and qualify as first authors.
dCorresponding Author: Rami Nasr, Division of Urology, Department of Surgery, American University of Beirut Medical Center, 11-0236/D50, Riad El-Solh 1107 2020, Beirut, Lebanon
Manuscript submitted November 30, 2019, accepted December 31, 2019
Short title: Inflammatory Markers in UTI
doi: https://doi.org/10.14740/wjnu398
| Abstract | ▴Top |
Background: Sepsis is a serious and life-threatening complication after transrectal ultrasound (TRUS)-guided prostate biopsy. It is critical to predict its occurrence prior to conducting the biopsy. The neutrophil/lymphocyte ratio (NLR) and platelet/lymphocyte (PLR) have been proven to be promising diagnostic indicators of infectious complications after various surgical procedures. The aim of this study was to determine whether inflammatory markers, NLR and PLR ratios, are useful surrogates to predict sepsis after TRUS biopsy.
Methods: A total of 378 patients underwent TRUS-guided prostate biopsy with no clinical evidence of prostatitis from December 2009 to May 2013 in American University of Beirut Medical Center. Data collected included age, smoking status, prostate size, post-void residual, prostate-specific antigen (PSA) value, blood culture, urine culture, Gleason score, pathology results, neutrophils, leukocyte count and platelet counts. Also, NLR and PLR were calculated. The primary outcome collected was sepsis. All data were entered and analyzed by Statistical Package for Social Sciences.
Results: Of the 378 patients who underwent TRUS biopsy, 31 patients developed sepsis. Septic patients were younger (63.7 ± 6.2 years) than non-septic patients (65.5 ± 8.0 years). No association between sepsis and NLR or PLR was observed; however, a significant association between sepsis and pre-biopsy urine analysis and pre-biopsy PSA (P = 0.011) was noted.
Conclusion: In this study, none of the pre-procedure NLR and PLR studied was found to predict sepsis after TRUS biopsy. Future efforts should be directed to investigate this relationship in prospective studies.
Keywords: Transrectal ultrasound-guided needle prostate biopsy; Sepsis; Blood inflammatory biomarkers
| Introduction | ▴Top |
Prostate cancer is the most frequently diagnosed cancer in men after middle age. In developed countries, the associated mortality rate ranks second to lung cancer [1]. Transrectal ultrasound (TRUS)-guided prostate biopsy is one of the most commonly performed urologic procedures in the United States and Europe, with approximately one million biopsies performed annually in each continent [2]. TRUS biopsy is a relatively safe procedure and chances of severe complications are low. Despite all preventive measures and improvement in techniques, the incidence of infectious complications has recently been rising [3]. Such complications prompt the urgent need for effective preventive strategies [4].
In an earlier study of 265 patients, the prevalence of sepsis was found to be 9.4% [5]. Antibiotic prophylaxis minimizes life-threatening septic complications [5]. However, septic complications cannot be avoided completely. For this reason, a more rational approach would be to identify potential pre-operative factors that would increase the risk of sepsis after biopsy. Inflammatory biomarkers are well studied in prostate cancer among other diseases. These biomarkers are proposed to act as an adjunct tool to improve the diagnosis of prostate cancer and predict the outcome and survival [6-9]; these biomarkers have been used as well to predict outcomes of inflammatory as well as infectious states [10, 11]. Multiple inflammatory markers such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are used to quantify the inflammation and thus could be used as predictors to infection. However, neutrophil/lymphocyte ratio (NLR) and platelet/lymphocyte ratio (PLR) are frugal parameters [12, 13] and are easily determined from complete blood count.
The aim of this study was to evaluate the prognostic impact of pre-biopsy NLR and PLR as predictors of febrile urinary tract infection (UTI) after TRUS biopsy in a tertiary medical care center.
| Materials and Methods | ▴Top |
Study design and setting
This was a retrospective study of patients who underwent TRUS biopsy from December 2009 until May 2013 at the American University of Beirut Medical Center, Lebanon after obtaining Institutional Review Board approval. Patients who had clinical evidence of infection before the procedure were excluded from the study. Patients eligible to be included in the study were those who underwent TRUS biopsy, and who had no clinical evidence of prostatitis or any other sign of infection and had negative urine culture prior to the procedure. Negative urine culture was defined as absence of bacteria. Moreover, patients with history of previous antibiotics treatment or patients on steroids were not included in the study. All patients underwent a standardized enema pre-procedure, peri-procedure antibiotic administration (aminoglycoside injection) and local anesthesia and Tru-Cut biopsy needle was used.
Patients’ medical charts were retrieved, and a database was compiled that included the following variables for each patient: age, smoking status, prostate size, post-void residual, prostate-specific antigen (PSA) value, blood culture, urine culture, urine analysis, Gleason score, pathology results, bowel preparation, peri-operative leukocyte count and platelet count. A urine analysis containing positive leukocyte esterase, nitrite, or white blood cell count (WBC) was considered positive and dealt with as a categorical variable. We noted the neutrophil and lymphocyte counts, and calculated NLR in addition to PLR. Finally, we traced all patients and identified those who developed sepsis, taking into consideration definition of sepsis which included hyper- or hypothermia, leukocytosis or leukopenia, positive blood culture, increased respiratory rate and hypotension.
Data entry and statistical analyses were performed using the Statistical Package for Social Sciences (SPSS), version 24.0. Descriptive analyses were conducted by calculating the mean and standard deviation (SD) for continuous variables and the number and percent for categorical variables. Associations between the different risk factors and the development of urosepsis were assessed using the Chi-square test for categorical variables or the Student’s t-test for continuous ones.
To account for the potential confounding effect of the different risk factors on the development of the outcome, analysis of variance was carried out. Multiple variables that could affect urinary infection after biopsy were included in the model. These variables included age, pre-biopsy PSA, pre-biopsy urine analysis and each one of the inflammatory parameters. Confidence interval (CI) of 95% was reported and P < 0.05 was considered significant. Receiver operating characteristic (ROC) curves were done to determine the best cut-off point for WBC, neutrophils, lymphocytes, platelets, NLR and PLR considering sepsis as a positive state variable.
| Results | ▴Top |
A total of 378 patients were included in the study. Mean age of patients is 61.3 years. Septic patients were relatively younger than non-septic patients (63.7 ± 6.2 years vs. 65.6 ± 8.0 years), respectively. A total of 31 patients out of 378 developed urinary tract sepsis.
Pre-biopsy variables were compared between septic and non-septic patients as shown in Table 1. The neutrophil, lymphocyte and platelet count were comparable between the two groups. As for NLR and PLR, the mean values for non-septic patients were 3.1 and 152.8, while for septic patients, values were 3.9 and 153.7, respectively. However, pre-operative PSA was significantly higher in those who had sepsis (40.7) versus those with no sepsis (11.0) (P = 0.011), and pre-biopsy positive urine analysis was significantly different between sepsis and non-sepsis groups (P = 0.011). Multivariate analyses showed that pre-operative urine analysis and pre-operative PSA were the only predictors of sepsis.
 Click to view | Table 1. Preoperative Variables in Septic Versus Non-Septic Patients |
ROC curves were constructed to determine a cut-off value for the NLR and PLR ratios. There was no cut-off point and the areas under the curves failed to show a relationship (Fig. 1).
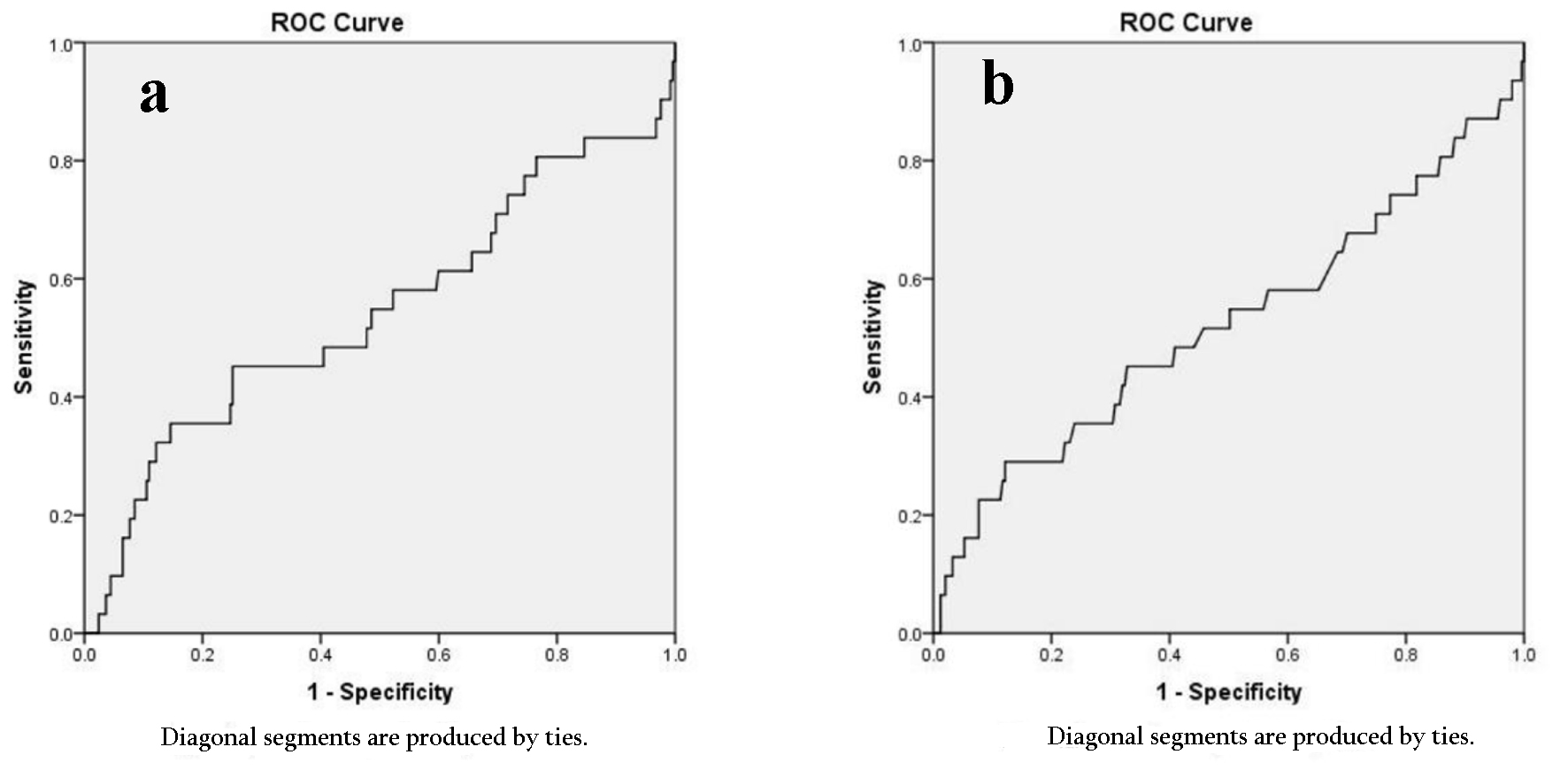 Click for large image | Figure 1. ROC curve. (a) Platelet lymphocyte ratio. (b) Neutrophil lymphocyte ratio. ROC: receiver operating characteristic. |
| Discussion | ▴Top |
In this study, we examined the role of NLR and PLR to predict febrile UTI after TRUS biopsy. Sepsis is defined as the presence of clinically or microbiologically documented infection in conjunction with systemic inflammatory response syndrome (SIRS). It is characterized by the presence of at least two of the following: body temperature more than 38 °C or less than 36 °C, heart rate more than 90 beats/min, respiratory rate more than 20 breaths/min or PaCO2 less than 32 mm Hg, WBC more than 12,000/µL or less than 4,000/µL or less than 10% detection of young neutrophils.
Multiple studies showed the association of high NLR and post-operative complications. A retrospective study exploring the association between NLR and wound healing in 101 diabetic patients showed that the ratio can be used as a surrogate of systemic inflammation, wherein higher NLRs were significantly associated with higher rates of non-healing of wounds (P = 0.01) [13]. Another study conducted on 298 pediatric patients with febrile UTI showed that NLR was elevated in the group of acute pyelonephritis with dimercaptosuccinic acid (DMSA) defect (P < 0.001) [12]. The authors found that NLR was an independent predictor for positive DMSA defect in UTI patients (P < 0.001) [12].
Another study found that NLR correlated with sequential organ dysfunction scores [14] and post-operative NLR was a significant predictor of complications after colorectal surgery in the univariate analysis. Patients with an NLR of 9.3 or above on the first day after elective colorectal surgery appear to have an increased risk of complications. Data have emerged on the ability of WBC subtypes, specifically the NLR to predict 30 days, 6 months and 12 months outcome after emergency abdominal surgery in the elderly [15] and to identify patients at increased risk of death within 2 years of/after major vascular surgery [16]. Another study highlighted the potential value of using NLR as a predictor of complications after hip fracture surgery, in which the authors found that NLR > 5 at day 5 after hip fracture surgery is associated with higher risk of post-operative mortality, cardiovascular complications/events and infections [16].
Since all studies tried to correlate NLR to post-operative complications and mortality, we tried to asses if this ratio can be used to predict who will be at higher risk of infectious complications after TRUS biopsy. NLR provides a simple index of the systemic inflammatory response and correlates with organ dysfunction scores and clinical course in critically ill patients [14]. Thus, NLR could provide a simple method to identify patients at high risk of post-operative complications. NLR is a simple index of the systemic inflammatory response.
In this study, we found that the two ratios failed to predict sepsis prior to the procedure even when controlling for other factors such as age, diabetes mellitus (DM), pathology results and pre-biopsy urine analysis. Both NLR and PLR values were not significant. There was a significant association between sepsis and pre-biopsy urine analysis (P = 0.011). There was no association between sepsis and the parameters even while adjusting for other variables in the multivariate analysis. PSA was associated with increased sepsis incidence in our cohort, which could be due to selection bias because a higher would render the patient less immune-competent from advanced disease; however, this is just a conjecture and needs to be further dissected in future studies. No definite studies have found a link between PSA level and risk of urinary sepsis after a TRUS biopsy. This is an observation that definitely needs re-affirmation with larger series and prospective studies.
To our knowledge, no previous studies have evaluated the value of pre-operative inflammatory ratios in predicting risk of post-TRUS biopsy infection-related complications. There are several limitations to our study. First, it is important to note the retrospective nature of the study and the small sample size. Second, we cannot preclude the role of NLR and PLR in predicting infectious complications after TRUS biopsy. Nevertheless, future prospective studies that look into the value of peri-operative novel inflammatory biomarkers are needed in attempt to predict patients who are at increased risk of infection-related complications. The authors acknowledge that the rate of urinary tract sepsis after biopsies is greater than that in the literature, which could be attributed to inherent retrospective selection bias and strict sepsis criteria implemented in the study.
Conclusion
Although rectal enema, potential antibiotics and prophylaxis prior to biopsy are commonly used, the rate of infection is still high. Thus, we need to identify biomarkers in order to stratify patients into low versus high risk for post-biopsy infection. Our study shows that NLR and PLR failed to predict sepsis after TRUS biopsy.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Not applicable.
Author Contributions
NAH and MS contributed to manuscript writing. RN and MS contributed with the idea. AY and NAH contributed with data collection and analysis.
| References | ▴Top |
- Torre LA, Siegel RL, Ward EM, Jemal A. Global cancer incidence and mortality rates and trends - an update. Cancer Epidemiol Biomarkers Prev. 2016;25(1):16-27.
doi pubmed - Loeb S, Carter HB, Berndt SI, Ricker W, Schaeffer EM. Complications after prostate biopsy: data from SEER-Medicare. J Urol. 2011;186(5):1830-1834.
doi pubmed - Nam RK, Saskin R, Lee Y, Liu Y, Law C, Klotz LH, Loblaw DA, et al. Increasing hospital admission rates for urological complications after transrectal ultrasound guided prostate biopsy. J Urol. 2010;183(3):963-968.
doi pubmed - Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
doi pubmed - Shahait M, Degheili J, El-Merhi F, Tamim H, Nasr R. Incidence of sepsis following transrectal ultrasound guided prostate biopsy at a tertiary-care medical center in Lebanon. Int Braz J Urol. 2016;42(1):60-68.
doi pubmed - Minardi D, Scartozzi M, Montesi L, Santoni M, Burattini L, Bianconi M, Lacetera V, et al. Neutrophil-to-lymphocyte ratio may be associated with the outcome in patients with prostate cancer. Springerplus. 2015;4:255.
doi pubmed - van Soest RJ, Templeton AJ, Vera-Badillo FE, Mercier F, Sonpavde G, Amir E, Tombal B, et al. Neutrophil-to-lymphocyte ratio as a prognostic biomarker for men with metastatic castration-resistant prostate cancer receiving first-line chemotherapy: data from two randomized phase III trials. Ann Oncol. 2015;26(4):743-749.
doi pubmed - Sonpavde G, Pond GR, Armstrong AJ, Clarke SJ, Vardy JL, Templeton AJ, Wang SL, et al. Prognostic impact of the neutrophil-to-lymphocyte ratio in men with metastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2014;12(5):317-324.
doi pubmed - Kawahara T, Fukui S, Sakamaki K, Ito Y, Ito H, Kobayashi N, Izumi K, et al. Neutrophil-to-lymphocyte ratio predicts prostatic carcinoma in men undergoing needle biopsy. Oncotarget. 2015;6(31):32169-32176.
doi pubmed - Abou Heidar N, Labban M, Bustros G, Nasr R. Inflammatory serum markers predicting spontaneous ureteral stone passage. Clin Exp Nephrol. 2019.
doi pubmed - Kaplan M, Ates I, Yuksel M, Arikan MF, Aydog G, Turhan N, Gokbulut V, et al. The role of the PLR-NLR combination in the prediction of the presence of Helicobacter pylori and its associated complications. Saudi J Gastroenterol. 2018;24(5):294-300.
doi pubmed - Han SY, Lee IR, Park SJ, Kim JH, Shin JI. Usefulness of neutrophil-lymphocyte ratio in young children with febrile urinary tract infection. Korean J Pediatr. 2016;59(3):139-144.
doi pubmed - Vatankhah N, Jahangiri Y, Landry GJ, McLafferty RB, Alkayed NJ, Moneta GL, Azarbal AF. Predictive value of neutrophil-to-lymphocyte ratio in diabetic wound healing. J Vasc Surg. 2017;65(2):478-483.
doi pubmed - Zahorec R. Ratio of neutrophil to lymphocyte counts - rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy. 2001;102(1):5-14.
- Vaughan-Shaw PG, Rees JR, King AT. Neutrophil lymphocyte ratio in outcome prediction after emergency abdominal surgery in the elderly. Int J Surg. 2012;10(3):157-162.
doi pubmed - Bhutta H, Agha R, Wong J, Tang TY, Wilson YG, Walsh SR. Neutrophil-lymphocyte ratio predicts medium-term survival following elective major vascular surgery: a cross-sectional study. Vasc Endovascular Surg. 2011;45(3):227-231.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
World Journal of Nephrology and Urology is published by Elmer Press Inc.
